
(13-4) is hypothetical for any components that are supercritical. (13-4) can be applied, the former is generally preferred because it involves only a single equation of stateĪpplicable to both phases and thus would seem to offer greater consistency. At low to moderate pressures, accurate prediction of the latter is crucial to the application of Eq. Where different equations of state may be used to predict the pure-component liquid fugacity coefficient Of and the vapor-mixture fugacity coefficient, and any one of a number of mixture free-energy models may be used to obtain the liquid activity coefficient y,L. Īn alternative K-value formulation that has received wide application to mixtures containing polar and/or nonpolar compounds is The Wong-Sandler mixing rules for cubic equations of state now permit such equations to be extended to mixtures of organic chemicals, as shown in a reformulated version by Orbey and Sandler. Similar results are achieved with the PR correlation. The ability of the SRK correlation to predict K values even when the pressure approaches the convergence pressure is shown for a multicomponent system in Fig. Computer programs for K values derived from the SRK, PR and other equations of state are widely available in all computer-aided process design and simulation programs. The Starling extension of the BWR equation (Fluid Thermodynamic Properties for Light Petroleum Systems, Gulf, Houston, 1973) predicts K values and enthalpies of the normal paraffins up through n-octane, as well as isobutane, isopen-tane, ethylene, propylene, nitrogen, carbon dioxide, and hydrogen sul-įide, including the cryogenic region. The SRK and PR equations belong to a family of so-called cubic equations of state.
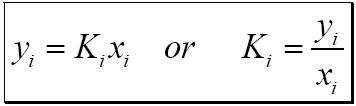
These equations include those of Bene dict-Webb-Rubin (BWR), Soave (SRK), who extended the remarkable Redlich-Kwong equation, and Peng-Robinson (PR). (13-3) have been restricted to mixtures of nonpolar compounds, namely, hydrocarbons and light gases. Until recently, equations of state that have been successfully applied to Eq. Consistent equations for enthalpy can similarly be derived. Where the mixture fugacity coefficients L for the liquid and ,V for the vapor are derived by classical thermodynamics from the PVT expression. When a single pressure-volume-temperature (PVT) equation of state is applicable to both vapor and liquid phases, the formulation used is Preferred analytical correlations are less empirical in nature and most often are theoretically based on one of two exact thermodynamic formulations, as derived in Sec. 13-14 K values (K = y/x) in light-hydrocarbon systems.
